Retatrutide Dosage Calculator: Understanding Clinical Titration
Explore the science behind the triple-agonist research compound and how clinical titration protocols are structured.
Learn About the CompoundRetatrutide is an experimental medication currently being studied for obesity and type 2 diabetes. As interest in this “triple agonist” grows, many people are looking for information on how it is administered. This Retatrutide dosage calculator is for educational purposes only; it is designed to help you understand the titration schedules used in clinical trials.
What is Retatrutide?
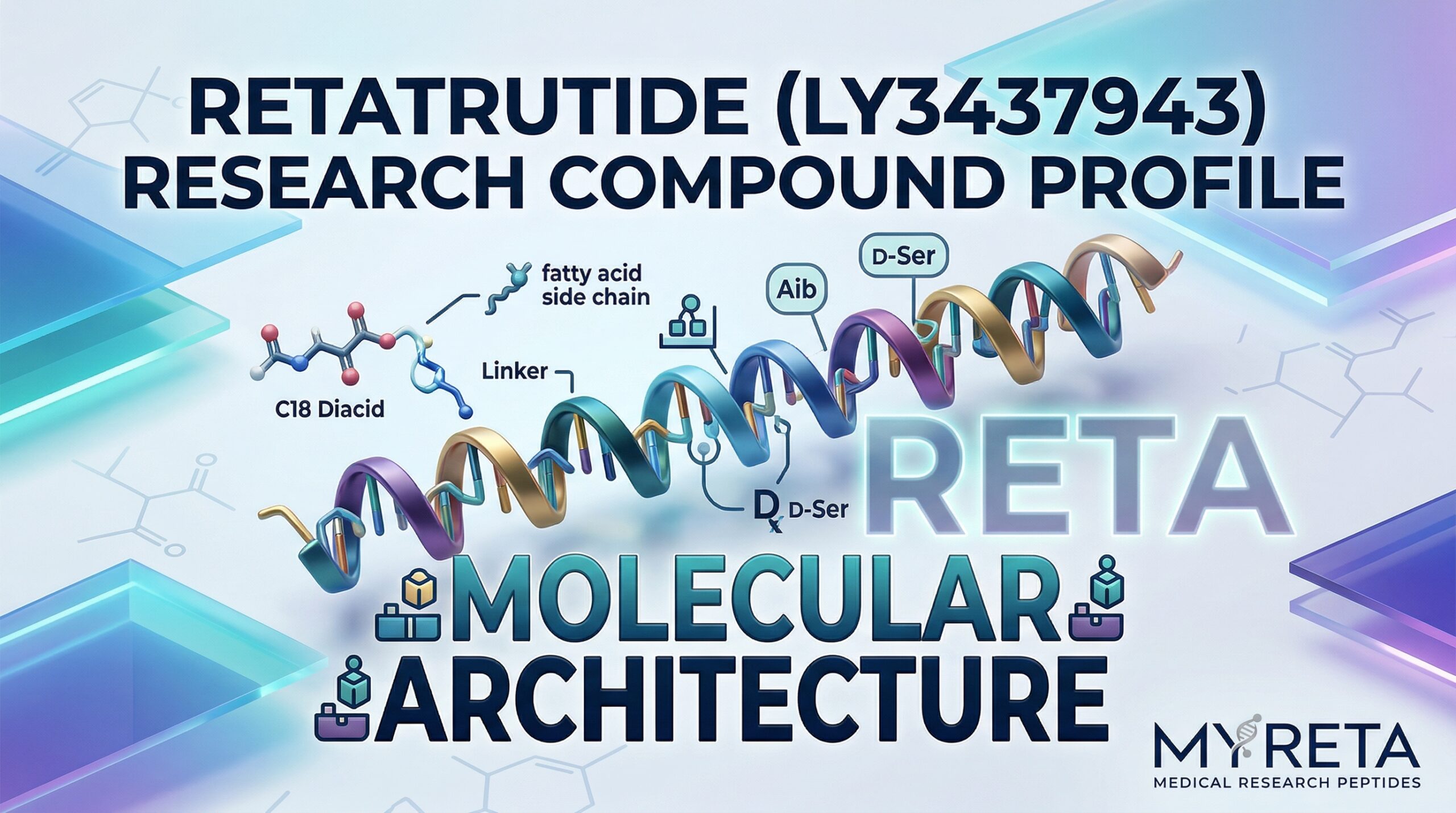
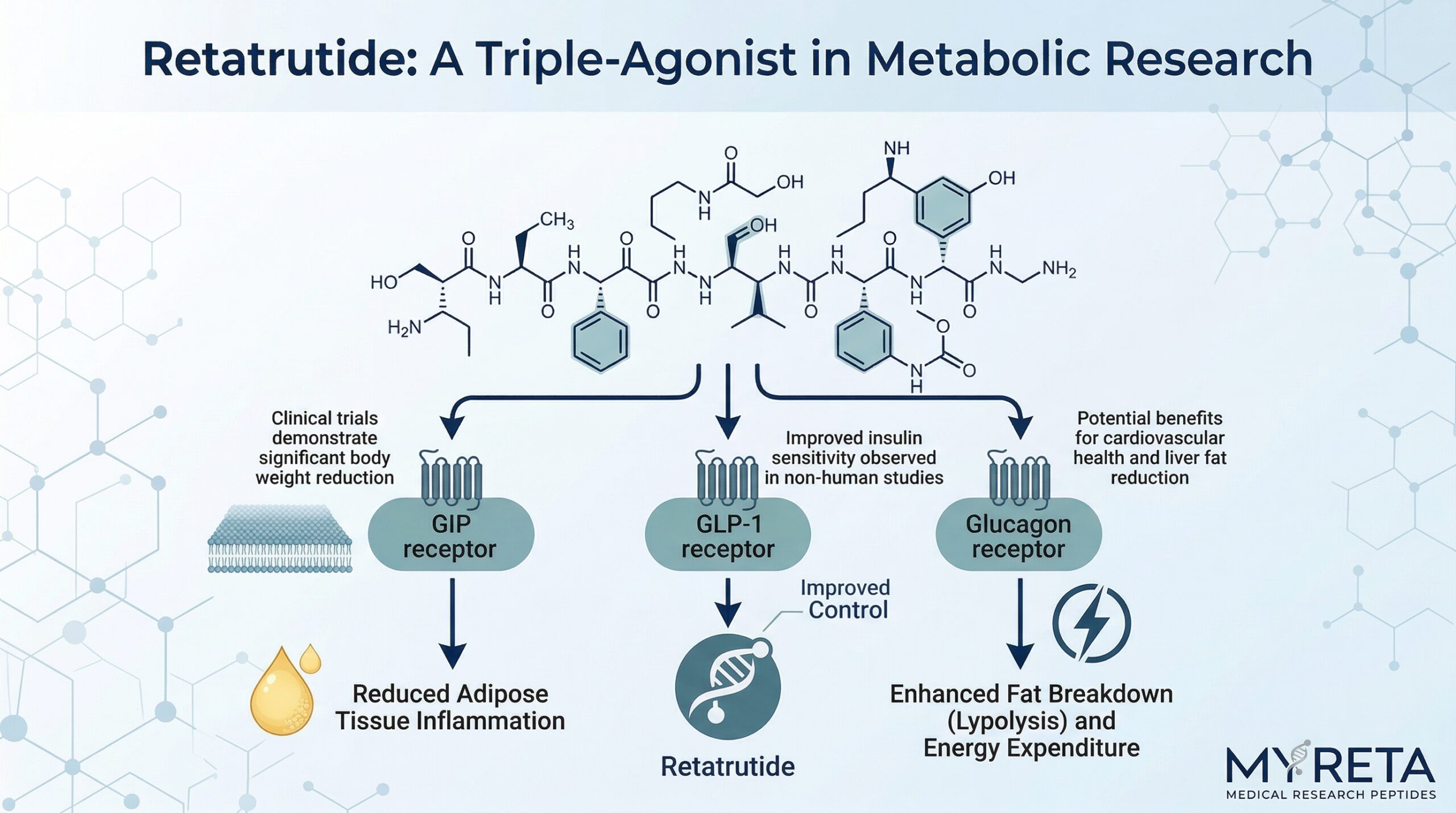
Retatrutide is a triple hormone receptor agonist. Unlike treatments that target one or two receptors, it activates three simultaneously:
- GLP-1 Receptor: Stimulates insulin secretion, suppresses glucagon, slows gastric emptying, and promotes feelings of fullness.
- GIP Receptor: Increases insulin secretion and affects energy expenditure and fat metabolism.
- Glucagon Receptor: Boosts energy expenditure and promotes fat oxidation.
Clinical Trial Data and Efficacy
Phase 2 trial results have made Retatrutide a major focus in metabolic medicine. In studies published in the New England Journal of Medicine, participants on the highest dose (12mg) lost an average of over 24% of their body weight after 48 weeks.
Dosage and Titration Protocols
In clinical trials, Retatrutide is given as a once-weekly subcutaneous injection. Because the body needs time to adjust, doctors use a “titration” process—starting at a low dose and gradually increasing it over several weeks.
- Starting Dose: Typically 2mg.
- Titration: Doses are increased incrementally based on individual tolerance.
- Maximum Dose: Clinical trials have tested doses up to 12mg.
Frequently Asked Questions
Is Retatrutide available for purchase?
No. Retatrutide is still in clinical development and is not available for public prescription or purchase.
How does the dosage calculator work?
The calculator provides an estimate based on published clinical trial titration schedules. It is meant to show how doctors manage dose escalation in a controlled setting. It does not account for your medical history or current health status.