Retatrutide: A Triple-Agonist in Metabolic Research
Exploring the potential of the Reta-class of triple hormone receptor agonists in metabolic research contexts.
Read Latest ResearchResearch into obesity and type 2 diabetes is evolving rapidly. Following the success of GLP-1 receptor agonists, pharmaceutical research has shifted toward more complex, multi-hormonal therapies. Eli Lilly’s retatrutide, an investigational triple hormone receptor agonist, is currently a focus of research in metabolic science.
Clinical data, including results from the March 2026 Phase 3 TRANSCEND-T2D-1 trial, suggests that retatrutide exhibits properties relevant to weight regulation and glycemic modulation in research models. By targeting the GIP, GLP-1, and glucagon receptors simultaneously, the molecule has demonstrated effects that warrant further scientific investigation.
The Science of the Triple Agonist
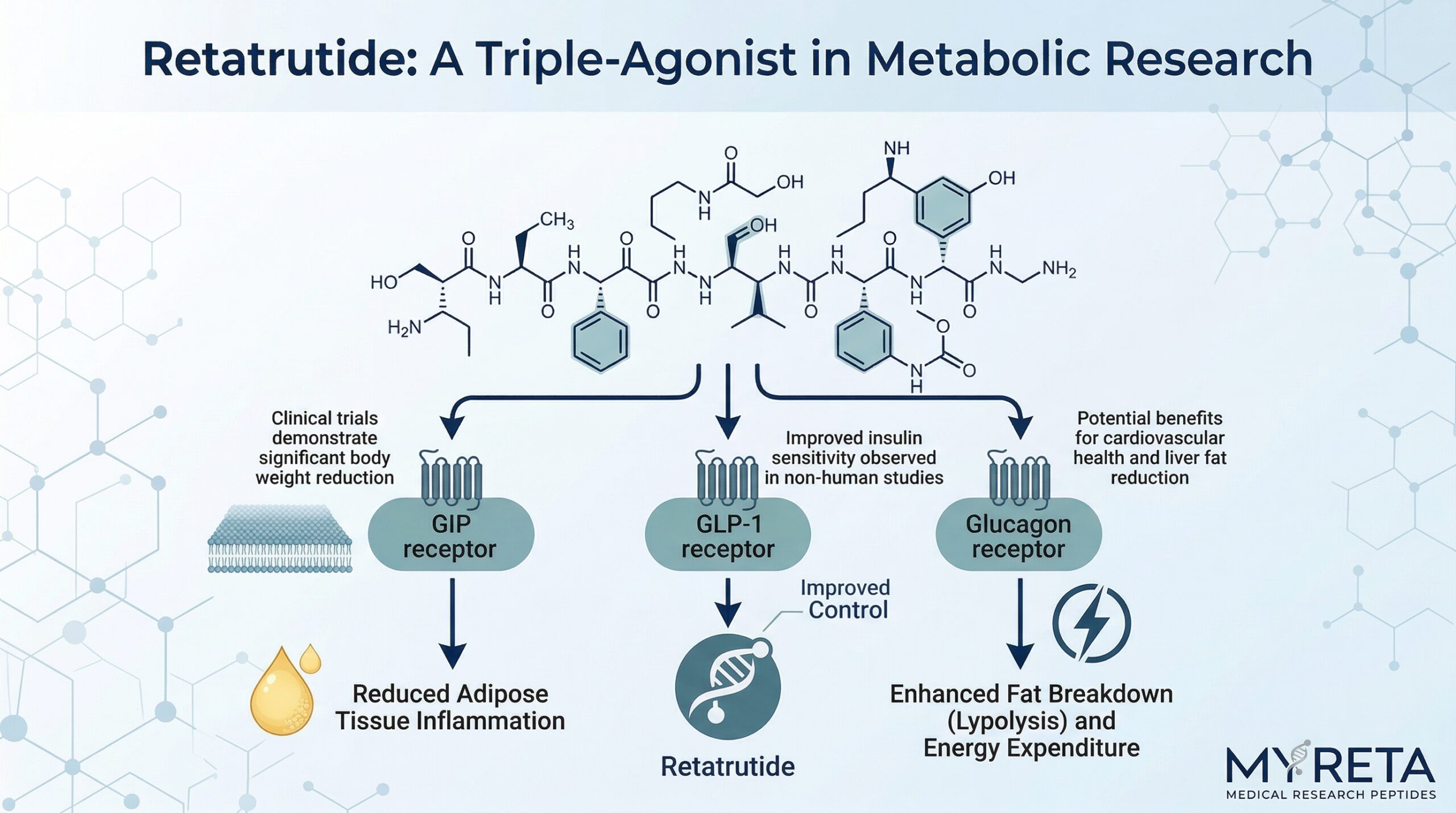
Retatrutide’s unique mechanism of action distinguishes it from other compounds studied for metabolic effects. While traditional GLP-1 agonists focus on a single pathway, retatrutide functions as a “triple agonist,” integrating signals from three distinct metabolic receptors:
- GLP-1: Associated with slowed gastric emptying, appetite modulation, and glucose-dependent insulin secretion.
- GIP: Associated with improved insulin release and regulation of food intake.
- Glucagon: Associated with increased energy expenditure and metabolic regulation, including the breakdown of stored energy.
Recent Developments and Clinical Trials
The investigation of retatrutide has been marked by consistent data across several key studies, positioning it as a subject of significant interest for future metabolic research.
- July 2023: Phase 2 trial results indicated observed effects relevant to obesity, type 2 diabetes, and metabolic dysfunction-associated steatotic liver disease (MASLD).
- March 2026: Eli Lilly announced topline results from the Phase 3 TRANSCEND-T2D-1 trial, reporting significant A1C reductions and weight loss in study subjects.
Regulatory Status
Retatrutide is not yet approved by regulatory bodies. It is currently an investigational compound restricted to clinical research settings. Access to such compounds is limited to legitimate, authorized research protocols.
Frequently Asked Questions
What is retatrutide?
Retatrutide is an investigational triple-hormone receptor agonist that mimics the biological effects of GLP-1, GIP, and glucagon, under investigation for its role in blood glucose regulation and weight modulation.
Comments
One response to “Retatrutide – What is Reta? – People who’ve seen the news about Phase 3 trial results wanting to learn more (informational)”
[…] Learn More About Reta […]